As the second wave of COVID is beginning to ebb, many in India are worrying about a future third wave. The best way we know to protect against the next wave is to vaccinate the population. In a previous post we explored (1) the progress being made to vaccinate India. In the this post two we explore (2) the supply of vaccines to the Centre and then states. In the next post we will examine (3) the administration of vaccine doses, including the problem of wastage of doses.
Before we jump in, we repeat here our main take-aways from these three posts:
India is vaccinating at a daily rate that is short of what would be required to meet its stated goal of vaccinating 20% of the population by July.
There is a lot of variation in how different states are performing. Interestingly, some Southern states that are typically higher performing lag behind other states.
India does not appear to have enough planned supply through July to meet its vaccination target.
The supply of vaccines from the Centre to states is mainly a function of their population. It does not appear to be affected by political circumstances.
Even though supplies are constrained, states with greater demand seem to administer a greater share of the doses they are provisioned.
States with greater demand have also have less wastage via expiration of doses. Interestingly, we find evidence that administration of Covaxin is associated with greater wastage, consistent with news stories that the larger 20-dose vials of Covaxin increase the risk of doses going unused before the vial expires.
This leaves us with the following three recommendations.
India should increase the production or procurement of vaccinations. If this is not feasible, it should explore whether solutions, like de-prioritizing those people who already have anti-COVID antibodies and fractional dosing, would be effective at increasing the value of limited vaccine doses.
There may be value in switching from allocation to states in proportion to population to allocation to states (or even districts) in proportion to future risk-based so that we allocate limited doses as impactfully as possible.
Even though supply is constrained, efforts to encourage demand (via marketing or lotteries) will reduce the risk of wastage. India may also want to consider making it easier to access doses by increasing the number of vaccination sessions it holds at each clinic.
The government should focus on improving rural distribution chains and building more durable adult vaccine infrastructure. Because the risk of future mutation may require future shots with reformulated vaccines, overcoming these logistical challenges should be viewed as a long-term investment in public health.
Going forward, we will work to keep people abreast of vaccination progress through our Twitter bot @covidmetrics.
II. The supply of vaccines to the country and from the centre to the states
To explore the causes of slow vaccine administration, we separate the problem into parts: supply to the centre, supply from the centre to the states, and then administration by states.
A. Supply to the Centre
The Government of India’s roadmap for vaccine production in India is presented in Table 1. (These numbers seem consistent with news stories such as this, this, this, this, this, this and this.) Assuming the quantities in the table are the number of doses rather than the number of people with 2 doses, then it seems India will not have adequate supply to vaccinate 6.5 million persons per day, i.e., 390,000 doses per month, until September, well past the July deadline. Even if only 1 dose were given per person, August would be the earliest India would get 195,000 doses per month required to hit the run rate necessary to meet the July target. It is possible there is unused supply in state inventories that would help, but it seems unlikely that is enough to hit MOH’s target.
Table 1. Government of India’s expected vaccine production/procurement schedule.
Production capacity for vaccines to be consumed in India is hobbled by limited supply of inputs into vaccines and by uncertainty regarding the value of investment in expanding production. Some of the inputs are simply limited, while others are limited by policies such as the Defense Production Act in the US. While the US has relaxed DPA enforcement so as to allow more exports of intermediate goods to foreign vaccine manufacturers, the slow growth in vaccine output suggests either supply chain and/or other obstacles remain.
At the same time, there may be hesitancy to expand production because outputs are subject to seizure by government or price controls and government marketing restrictions, each of which reduce return. This is not a problem unique to India. The same concern arises with discussions of price controls or collective bargaining on prices in the US and Europe. However it is not a problem remote to India either. For example, if the Serum Institute for India has to abrogate contracts with foreign countries, then those countries will not rely on SII. If they do not rely on SII, then SII can expect lower returns from investment in capacity. The unclear regulatory status of vaccines, in light of things like blood clot side effects from the AstraZeneca (AZ) vaccine or the lack of final clinical trial results on COVAXIN, compounds the risk to returns.
While India has begun to seek doses from outside India, this avenue is unlikely to substantially ease supply constraints. For example, India has tried to access the stockpile of 60 million AZ vaccines sitting in Baltimore, USA. But we have heard that, because AZ is not approved in the US, the FDA still has to review safety and perhaps efficacy before these doses can be released. Nor is it clear those doses will go to India rather than US neighbors such as Mexico and Canada. India has begun discussing procurement for Pfizer doses since new came out that that vaccine may not require -80 degree Celsius storage. However, no breakthroughs have been achieved yet. In any case, 60 million AZ doses and future capacity for Pfizer is unlikely to cover the hundreds of millions that remain to be vaccinated in India.
One might ask whether IP rights are an obstacle. Some have lauded the US announcement that it is open to removing IP rights on COVID vaccines. (This followed a filing in October 2020 by India and South Africa calling for waiver of IP rights for COVID-related pharmaceuticals.) We are unconvinced that these moves represent a serious shift in IP status. There are other nations that would have to approve the waiver (and perhaps even US courts). The Trade-Related Aspects of Intellectual Protection (TRIPS) Agreement may already have provisions to allow waivers for public health. Finally, as production agreements the GOI table above suggest, many vaccine IP holders are willing to lease rights to Indian manufacturers.
Even were the US announcement a meaningful change in IP status for COVID vaccines, giving Indian (and other foreign) manufacturers the right to produce vaccines does not overcome supply chain problems or price uncertainty. It is even less likely to help new entrants. Vaccine manufacturing requires skilled personnel, who are in short supply globally. It also requires production techniques and careful quality control processes, which take time to build. After the pandemic, we should build out a global vaccine supply chain, both for future pandemic and also for the boom in vaccine uses that mRNA technology portends. But in the short-run, the world has a limited ability to dramatically scale production simply with IP law changes.
In short, India is likely to experience tight supply, relative to its stated vaccination targets and to more ambitious, but public health-appropriate targets, like vaccinating its entire adult population this year or even before a third wave.
Therefore, it is important to couple efforts to increase manufacturing with efforts to economize existing capacity. A useful analogy is energy conservation and efficiency. Energy shortages or price jumps require countries to make use of that energy more efficiently and to allocate limited energy efficiently across sectors. Likewise, we need to make existing doses cover more persons and to prioritize which persons get vaccinated first.
One helpful effort to increase efficiency, i.e., the number of people vaccinated by each bottle, is the government’s decision to increase the spacing between 1st and 2nd doses to 12 to 16 weeks. This spacing will provide some short term relief from limited vaccine supplies. But whether that spacing will really help or just defer the problem of supply depends on whether the government adequately scales production before delayed 2nd doses come due.
Another boost to efficiency would be to attempt to screen out or delay vaccination for those with natural immunity from a prior COVID infection. A simple but useful model to compare prior infection with vaccination is as follows: A first infection for a susceptible person is equivalent to a first dose of vaccine for that person. Thus a vaccine dose for a previously infected person is like the second dose of vaccine for a susceptible person. Given how large the first and second waves were, it is possible a much higher fraction of the country has received a de facto first dose than vaccine statistics indicate. In India could both delay first jabs for previously infected or avoid a second jab for that group, it could cover more people with its existing stock of vaccines.
The problem is identifying who was previously infected. Serological surveys will help, but they are not a panacea. First, we are confident antibody tests can identify infection in the last 3-4 months. Beyond that range, we are uncertain they can because anti-COVID antibodies may fade from the blood a few months after the infection is cleared. A silver lining of the second wave is that a large percentage of the population may have antibodies that can be detected by even myopic antibody tests. Second, antibody tests are expensive, costing as much as a vaccine in some cases. Nevertheless, they are a way to focus limited supplies of vaccines on not just those that are greatest health risk from infection, but those for whom the vaccine is likely to make a bigger contribution to immunity.
A third, potential strategy to increase efficiency of existing vaccine stock is fractional dosing. We know that fractional dosing works for some vaccines, such as Pfizers. The reason is that Pfizer, when testing the vaccine, picked a dose that was so large it would be very confident that it worked and passed regulatory hurdles. Thus the dosage chosen was not what was minimally adequate, but what minimized approval risk. Fortunately, smaller doses are substantially as effective as larger doses. Thus, the same amount of vaccine quantity as measured in ml can go further than we thought before. The problem for India is that fractional dosing studies have not been conducted for the two main vaccines used there (Astra Zeneca or COVISHIELD and COVAXIN). These should be done post haste!
We conclude with one small point. The Centre recently reclaimed full control over government procurement of vaccines. If this is followed by centralization of vaccine allocation, it will mean that all efforts to experiment with stretching and optimally allocate doses have to be addressed to this Centre. This makes the stakes of these decisions very high. If the decisions are correct, they are correct for the entire country. But if they are incorrect, they are incorrect for the entire country. In addition, it is easier to experiment at the state level than at the Centre because, within any government unit, there is pressure to be uniform in allocation and smaller state decisions are less likely to attract pre-judged media coverage. So it may be harder for the Centre to try a pilot in just 1 area on, e.g., screening for antibodies before vaccinating. If one thinks experimentation is important (and we do), then state-level allocation have some merit.
B. Supply by the Centre to the states
The primary determinant of how vaccinates are allocated by the Centre to the states is population. As Figure 5 shows, larger states get more doses. On average, states have gotten 12 million doses for each 100 million persons in the population. (The actual rate is likely lower since we are using 2011 population data and the population has surely grown.)
Figure 5: Vaccine Supply and Population
Source: Ministry of Health and Family Welfare, Press Release and Census 2011
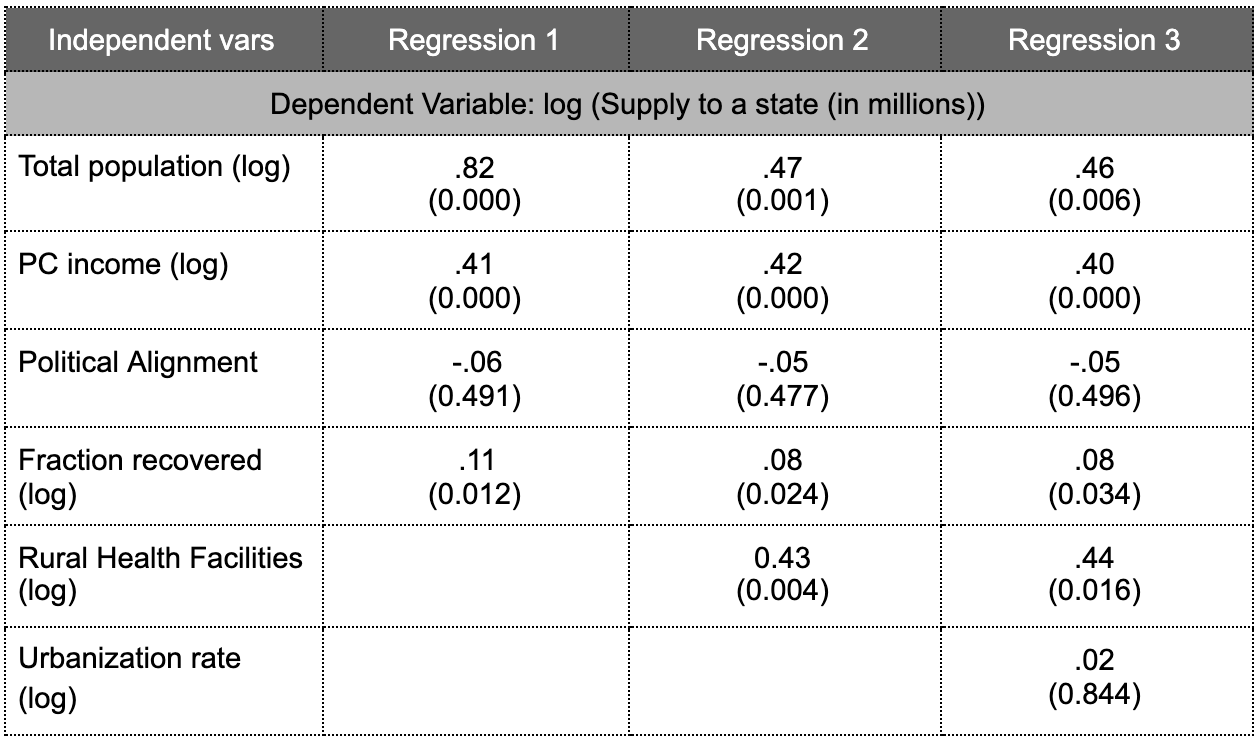
There are some outliers, such as UP and Bihar on the low end, and Maharashtra, Gujarat and Rajasthan on the high end. We explored this variation via linear regression analysis. Beyond population, we looked at other possible predictors of supply to states, including wealth, politics, COVID risk, and logistical capacity. Our specification regresses log supply on log explanatory variables so that coefficients are comparable in percent-change units. We find that, aside from population, per capita income, COVID risk and logistics best predict supply (Tables 1A and 1B).
By far the best predictor of supply is population (Table 2). A 10% increase in state population is associated with roughly 4.5% greater supply from the Centre. This is consistent with stated MOHFW policy on vaccine allocation.
After population, rural logistics, which we measure via rural health facilities, is a strong predictor of supply from the Centre. A 10% increase in rural facilities is associated with a 4.3% increase in supply. This suggests that the Centre is implicitly considering rural logistic capacity in deciding where to supply doses first. This is important because supply without distribution capacity may lead to greater wastage.
After population, per capita income is a strong predictor. A state with 10% greater per capita income receives roughly 4% greater supply. One might wonder if the relationship of supply with per capita income reflects the fact that wealthier states have been able to procure more doses directly from vaccine manufacturers. However, we do not believe that the quantity procured directly states is likely to be a meaningful portion of total supply to date. (Please let us know if you think we are wrong.) Our tentative conclusion, then, is that richer states appear to be receiving more supply. Although it is speculative, perhaps the Centre either implicitly values economic performance in vaccine allocation or perceives wealthier states to have better ability to administer doses quickly.
Prior COVID exposure also correlates with vaccine supply, though less strongly. A 10% increase in the fraction of the population that has previously been infected with COVID is associated with a 8% increase in supply. (We approximate the population that was previously infected by (1) scaling up confirmed cases by serological data on the fraction that have antibodies at a given point in time and (2) taking the cumulative sum of this adjusted confirmed case curve.) This suggests that, to some extent, the Centre is also paying attention to COVID burden. The only flag we would raise is that the Centre may not want to allocate doses in positive proportion to the fraction previously infected. When a high fraction have natural immunity, the remaining number at risk is lower. A better approach is to calculate a constant hazard rate based on this natural immunity level, a relatively simple calculation; this would suggest a non-monotonic relationship between fraction infected and future risk.
Some of the non-results are also reassuring. Some may be worried that political alignment with the Centre affects allocations. This does not appear to be true.
Table 2. Relationship between vaccine supply to different states and population, income, political alignment, COVID risk, and distribution capacity.
References
Asher , S., Lunt , T., Matsuura , R., & Novosad, P. (2021). DDL COVID India. Retrieved from Data Development Lab: http://www.devdatalab.org/
covid19india.org. (2021). Retrieved from covid19india.org
Government of India. (2021). Co-WIn. Retrieved from Dashboard: https://dashboard.cowin.gov.in
IDFC Institute. (2021). The Indian OVID-19 Alliance, TICA. Retrieved from https://www.idfcinstitute.org/projects/state-capacity/the-indian-covid-19-alliance-tica/
Ministry of Health and Family Welfare. (2020, October). Press Release: Dr Harsh Vardhan interacts with social media users during Sunday Samvaad-4. New Delhi.
Ministry of Health and Family Welfare. (2021, May 11). More than 18 crore vaccine doses provided to States/UTs Free of Cost by Govt. of India, so far. Delhi.
Ministry of Health and Family Welfare. (2021, May 17). More than 20 crore vaccine doses provided to States/UTs Free of Cost by Govt. of India, so far . Delhi.
Office of the Registrar General & Census Commissioner, India. (n.d.). 2011 Census Data. Retrieved from Census India: https://censusindia.gov.in/2011-common/censusdata2011.html